Personal collections


In a hydrogen atom, the first six energy states of the electron have the following values:
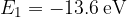 ,
, 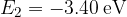 ,
, 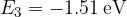 ,
, 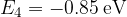 ,
, 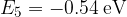 ,
, 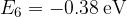
Calculate the maximum and minimum wavelength and frequency of the emitted light at the transition of the electron between the given energy states.