

In the chapter, Electric charge, we observed the forces between electrically charged bodies. We have seen that two bodies, one of which has a positive and the other a negative charge, attract each other. If both bodies have a positive charge or both have a negative charge, they repel each other.
Electric forces act at a distance, without direct contact between charged bodies. They are made possible by the electric field surrounding the charged bodies. An electric field is a "carrier of forces" between electrically charged bodies. We will learn more about the electric field in the chapter, Electric field strength.
In this chapter, we will learn about Coulomb's law. With the help of Coulomb's law, we will calculate the forces between two or more electrically charged bodies. If we imagine that the bodies are infinitely small, we call them point charges. So we will calculate the forces between point charges.
French physicist C.A. Coulomb was the first to investigate the forces between electrically charged bodies in the 18th century. He took two charged balls and measured the electric forces between them. With the help of precise measurements, he arrived at the result that the force between two charges is proportional to the magnitude of the two charges and inversely proportional to the square of the distance between them:
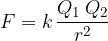
The constant 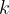 is called the Coulomb constant and is given as:
is called the Coulomb constant and is given as:
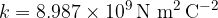
Today we know it in the form:
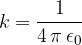
The electric force between two point charges is therefore given as:
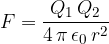
where 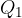 and
and 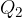 are the magnitudes of the charges,
are the magnitudes of the charges,  is the distance between the point charges and
is the distance between the point charges and 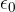 is the permittivity of free space which is given as:
is the permittivity of free space which is given as:
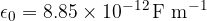
If we have several point charges in space, the total force is obtained by adding the forces of the charges vectorially:
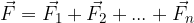
The electric force 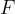 between two charged particles is calculated using Coulomb's law which is given as:
between two charged particles is calculated using Coulomb's law which is given as:
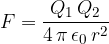
where the permittivity of free space 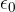 is given as:
is given as:
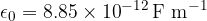
If a force from several charges acts on a specific charge, the forces are added vectorially:
The force is 0.72 mN. Because the charges are opposite, the force is attractive. It is directed towards the second charge.