Personal collections


State what is meant by an ideal gas.
Two cylinders A and B are connected by a tube of negligible volume, as shown in the figure below.
Initially, tap T is closed. The cylinders contain an ideal gas at different pressures.
Cylinder A has a constant volume of 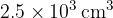 and contains gas at pressure
and contains gas at pressure 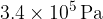 and temperature
and temperature 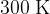 . Show that cylinder A contains
. Show that cylinder A contains 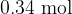 of gas.
of gas.
Cylinder B has a constant volume of 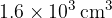 and contains
and contains 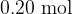 of gas. When tap T is opened, the pressure of the gas in both cylinders is
of gas. When tap T is opened, the pressure of the gas in both cylinders is 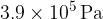 . No thermal energy enters or leaves the gas. Determine the final temperature of the gas.
. No thermal energy enters or leaves the gas. Determine the final temperature of the gas.
By reference to work done and change in internal energy, suggest why the temperature of the gas in cylinder A has changed.