Personal collections


Two cylinders X and Y each contain a sample of an ideal gas. The samples are in thermal equilibrium with each other. X has a volume of 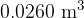 and contains
and contains 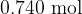 of gas at a pressure of
of gas at a pressure of 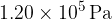 . Y has a volume of
. Y has a volume of 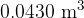 and contains gas at a pressure of
and contains gas at a pressure of 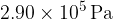 . Data for the two cylinders are shown in the figure below.
. Data for the two cylinders are shown in the figure below.
Show that the temperature of the gas in X is 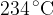 .
.
Determine the number 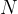 of molecules of the gas in Y. Explain your reasoning.
of molecules of the gas in Y. Explain your reasoning.