Personal collections


A litre of water at a temperature of 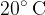 and 200 g of iron heated to a temperature of
and 200 g of iron heated to a temperature of 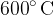 are placed in a thermally insulated container until a thermal equilibrium is established at a final temperature of
are placed in a thermally insulated container until a thermal equilibrium is established at a final temperature of 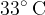 ? The specific heat capacity of water is 4200 J/Kg K. What is the specific heat capacity of iron?
? The specific heat capacity of water is 4200 J/Kg K. What is the specific heat capacity of iron?